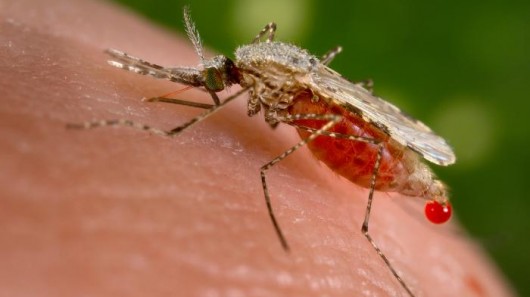
According to a new scientific publication, genetically engineered mosquitoes produced by the biotech company Oxitec (Intrexon) have escaped human control after trials in Brazil. They are now spreading in the environment, GM Watch reported Wednesday.
Source: GM Watch
The yellow fever mosquitos (Aedes aegypti) are genetically engineered to make it impossible for their offspring to survive. After release they were supposed to mate with female mosquitos of the species which transmit infectious diseases, such as Dengue fever, to diminish the natural populations.
However, the now published research shows that many offspring of the genetically engineered mosquitos actually survived and are spreading and propagating further. According to the scientists, between 10-60 percent of the mosquitoes in the region concerned are inheriting parts of the genome of the mosquitoes released in the trials. These findings are also confirmed in neighbouring regions where no such trials were conducted.
The long-term consequences for the transmission of diseases, the number of insects and the interactions with the environment cannot be predicted. The mosquitoes used for the genetic manipulation are originally from Cuba and Mexico. These insects used in the laboratory have now mixed with the Brazilian insects to become a robust population which can persist in the environment over a longer period of time. They might replace the original insects in the long term and even enhance the problems associated with the mosquitoes.
Christoph Then for Testbiotech commented, “The Oxitec trials have led to a situation that is largely out of control. The company has released its patented insects although it was known before that some insects could survive in the environment. The expectations of their investors were more important than the protection of health and the environment. There is no insurance and no fast-track mechanism to prevent severe damage in a worst-case scenario.
“This incident must have consequences for further applications of genetic engineering. Preventing the spread of genetically engineered organisms within natural populations has to become a priority.”
Oxitec misled the public
Oxitec misled the public and regulators regarding the GM mosquitoes, which it falsely described as “self-limiting”. It obtained funding from the Bill & Melinda Gates Foundation to develop the GM insects.
The new paper also reminds us that Oxitec’s strategy to reduce the population of mosquitoes did not work. It cites previous research as showing that the effectiveness of the release program began to break down after about 18 months: the population which had been suppressed rebounded to nearly pre-release levels.
Several research institutions players are planning to create genetically engineered trees, bees, corals, several insect species and other organisms that can spread in natural populations. The long-term impacts of such trials are unpredictable. Unintended effects can disturb or disrupt ecosystems and lead to the extinction of species. Therefore Testbiotech is proposing to introduce new criteria for the risk assessment of genetically engineered organisms to make sure they can be prevented from spreading uncontrolled into the environment.
The citation and abstract of the new publication is below.
Main source for commentary: Testbiotech
www.testbiotech.org/en/press-release/genetically-engineered-mosquitoes-out-of-control
—
Transgenic Aedes aegypti Mosquitoes Transfer Genes into a Natural Population
Benjamin R. Evans, Panayiota Kotsakiozi, Andre Luis Costa-da-Silva, Rafaella Sayuri Ioshino, Luiza Garziera, Michele C. Pedrosa, Aldo Malavasi, Jair F. Virginio, Margareth L. Capurro & Jeffrey R. Powell
Scientific Reports volume 9, Article number: 13047 (2019)
www.nature.com/articles/s41598-019-49660-6
Abstract
In an attempt to control the mosquito-borne diseases yellow fever, dengue, chikungunya, and Zika fevers, a strain of transgenically modified Aedes aegypti mosquitoes containing a dominant lethal gene has been developed by a commercial company, Oxitec Ltd. If lethality is complete, releasing this strain should only reduce population size and not affect the genetics of the target populations. Approximately 450 thousand males of this strain were released each week for 27 months in Jacobina, Bahia, Brazil. We genotyped the release strain and the target Jacobina population before releases began for >21,000 single nucleotide polymorphisms (SNPs). Genetic sampling from the target population six, 12, and 27–30 months after releases commenced provides clear evidence that portions of the transgenic strain genome have been incorporated into the target population. Evidently, rare viable hybrid offspring between the release strain and the Jacobina population are sufficiently robust to be able to reproduce in nature. The release strain was developed using a strain originally from Cuba, then outcrossed to a Mexican population. Thus, Jacobina Ae. aegypti are now a mix of three populations. It is unclear how this may affect disease transmission or affect other efforts to control these dangerous vectors. These results highlight the importance of having in place a genetic monitoring program during such releases to detect un-anticipated outcomes.



















Don’t fuck with mother nature you fucking carps. You are not god nor the creator of such.
will it not spread to the other animals