GeneWatch UK today warned that open releases of millions of GM mosquitoes produced by UK company Oxitec should not take place in Panama until members of the public have been fully informed about the risks.
GeneWatch also released a draft risk assessment provided by Oxitec to the US Department of Agriculture in 2011, which has not previously been published, showing that the company itself admits some of these risks (1). These include the risks that more invasive Asian Tiger (Aedes albopictus) mosquitoes move into the area, and that the number of cases of potentially fatal Dengue Haemorraghic Fever (DHF) could possibly increase.
The experiments in Panama are due to start on 15th February in Nuevo Chorillo, in the Arraiján district of Panama. The experiments have been approved by the Ministry of Health but no information about the possible downsides of the experiments has been provided to members of the public.
“Local people in Panama must be asked for their fully informed consent before these experiments begin” said Dr Helen Wallace, Director of GeneWatch UK, “This means the risks must not be hidden by the company. People must be able to discuss the pros and cons of these experiments and have a right to have their say”.
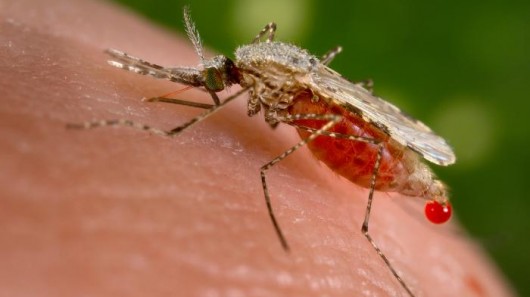
Oxitec’s GM Aedes aegypti mosquitoes have been genetically programmed to die at the larval stage. They are bred in the lab in the presence of an antidote to the genetic killing mechanism (the common antibiotic tetracycline), then vast numbers of males (millions for an experimental release or billions for a commercial one) are released into the environment so they outnumber the wild males and mate with wild females. Because most of the offspring die before adulthood, this is intended to reduce the wild population of Aedes aegypti mosquitoes, which carry the tropical disease dengue fever.
Oxitec conducted its first open release experiments of GM mosquitoes in the Cayman Islands in 2009-10 followed by a smaller experiment in Malaysia in 2010-11. Both countries have halted further releases. Larger-scale experiments began in Brazil in February 2011 and are still ongoing. Despite repeated claims of success made by Oxitec in the press, no results from the Brazil experiments have yet been published in scientific journals (2). Impacts on dengue fever have not been measured or reported. Experiments in the USA (Florida Keys) have been proposed but have not yet been approved by the US FDA.
“Aedes aegypti mosquitoes are part of a complex system which includes other mosquito species, the viruses they carry, and the humans that they bite” said Dr Wallace, “Local people should be aware that releasing large numbers of GM mosquitoes can pose risks to their health and the environment. They also need to know who will be liable if anything goes wrong – will Oxitec take responsibility for any problems, or just walk away?”
In its warning today, GeneWatch UK drew attention to the risks that should be considered and that local people should be told about (3). Some of these risks are acknowledged in a draft risk assessment submitted by Oxitec to the USDA, which GeneWatch has also released online today (1).
The major issues are:
1. Impact on mosquito populations. Oxitec has not assessed the possibility that mosquito numbers in areas neighbouring the trials could increase as a result of the experiments, or considered the serious risk that another species of dengue-transmitting mosquito, Aedes albopictus (the Asian Tiger mosquito), increases in numbers if the numbers of Aedes aegypti decrease. Researchers at Panama University have described Aedes albopictus as more dangerous than Aedes aegypti and it is regarded as a more invasive species which may be very difficult to tackle if it moves into the area (4). In its draft risk assessment submitted to regulators in the USA Oxitec states (5): “It is not clear to what extent Ae. albopictus could or would expand its range into areas currently dominated by Ae. aegypti but it is reasonable to expect a degree of such expansion if no countervailing activities are undertaken”.
2. Impact on dengue fever. Oxitec has published no evidence that population suppression of Aedes aegypti mosquito numbers in its target area will actually reduce the incidence or severity of dengue fever or the more severe form of the disease dengue hemorrhagic fever (DHF). In its draft risk assessment submitted to regulators in the USA Oxitec states (6): “It has been suggested that, in countries with very high transmission rates, reduction in transmission could increase the frequency of dengue hemorrhagic fever (DHF) even while decreasing the incidence of dengue fever”. In its draft risk assessment for the USA, Oxitec then dismisses this concern by making an unproven claim that the reduction in transmission will be well below the necessary level and pointing out that this concern is not relevant to the USA (where dengue fever is not endemic). However, this risk is highly relevant in Panama.
3. Release of biting females. Oxitec has recently published figures on the number of biting female GM mosquitoes that are inadvertently released (7). If correct, this would mean that 200 biting female GM mosquitoes are released in every million males, meaning that some people will inevitably be bitten. Current production of Oxitec’s GM mosquitoes in Brazil is 4 million a week. In the Cayman Islands, mechanical sorting was less effective, leading to about 5,000 biting female mosquitoes in every million males.
4. Survival and spread of GM mosquitoes. In the laboratory, 3% of the offspring of Oxitec’s GM mosquitoes survive to adulthood, even in the absence of the antidote tetracycline. This percentage could increase as resistance to the genetic killing mechanism evolves over time. When GM mosquitoes were fed cat food containing industrially farmed chicken, which contains the antibiotic tetracycline, the survival rate increased to 18% (8). A number of studies have found thatAedes aegypti mosquitoes can breed in septic tanks where there are high levels of contamination with antibiotics such as tetracycline. They also commonly live in areas where discarded takeaways are likely to contain meat contaminated with tetracycline. Increased survival rates would reduce the effectiveness of any population suppression effect over time, increase the number of biting GM females, and potentially allow the GM mosquitoes to establish in the wild.
5. Transfer of other traits to wild mosquitoes. When Oxitec’s GM mosquitoes breed with wild mosquitoes some of their other genetic characteristics will be passed on to the local wild mosquito population. Different strains of the same species are found in different places and some strains are more resistant to insecticides than others or better transmitters of disease. Oxitec’s GM mosquitoes are not developed from a native strain so the possible introduction of such traits needs to be considered. There are four different types of dengue virus and the harm to people’s health can be increased if some types can be transmitted more easily by the introduced strain than they were by the wild species already in the area. Other viruses, such as Yellow Fever, are also transmitted by Aedes aegypti mosquitoes and the impact of the new strain on transmission of such viruses also needs to be considered.
Notes:
(1) Oxitec draft environmental assessment. February 2011. On: www.genewatch.org/f03c6d66a9b354535738483c1c3d49e4/Environmental_Assessment.pdf
(2) Oxitec frequently claims its experiments have been successful at suppressing the wild Aedes aegypti mosquito population but it has only published the results of one population suppression trial (in the Cayman Islands) in the scientific literature. These trials were unsuccessful until Oxitec significantly increased the numbers of GM mosquitoes released and also placed GM mosquito larvae at regular intervals across the trial site. In Brazil, Oxitec’s experiments in Iteraba, Juazeiro required 54 GM to one wild mosquito to be released, a sign that the technology is ineffective. More recent experiments in Mandacaru took place only when very low numbers of mosquitoes are present in the dry season.
(3) Genetically Modified Mosquitoes: Ongoing Concerns. By Helen Wallace. 2013. TWN Biotechnology & Biosafety Series 15.twnside.org.sg/title2/biosafety/pdf/bio15.pdf
(4) www.panamaamerica.com.pa/notas/1103790-mosquito-aedes-albopictus-es-mas-peligroso-que-el-aegypti
(5) Page 25 of Ref (1).
(6) Page 72 of Ref (1).
(7) www.jove.com/video/3579/mass-production-genetically-modified-aedes-aegypti-for-field-releases Reports female contamination on average is 0.02%.
(8) Oxitec originally hid this information (see: www.genewatch.org/article.shtml?als%5Bcid%5D=492860&als%5Bitemid%5D=569476 ) but later admitted to an 18% survival rate of larvae fed on cat food in a published paper: www.ncbi.nlm.nih.gov/pmc/articles/PMC3653897/ . The company still argues that Aedes aegypti will not encounter sufficient levels of tetracycline in the environment, although industrially farmed chicken is widespread and Aedes aegypti also breed in septic tanks where there are likely to be high levels of antibiotics.


















Holy Cow, when will it stop? This has gone beyond ridiculous. An extremely reckless roll of the dice.
This is crazy! I live in Chame district and don’t want those things near me. How can we put a stop to this!?
Please contact the Ministry of Health in Panama. They are the Ministry who have approved the trial.
I would like you to try to see if anyone will answer to you in the way you think they will, they are useless believe me the money is what they worry about and they get paid a lot to approve whatever the investors want to approve.
Wow! Although I applaud the effort on doing something about native mosquito populations i wonder how they came to the conclusion that Arraijan is the right spot? Its fairly populated with a huge mall that recently opened.
how do they control the situation if something has to be done?
Since most mosquitoes have a very short range territory They should conduct that experiment on an island where they have a better chance to regain control if anything goes wrong.
the thing about messing with nature is that no matter how many models they run on a computer they only are an approximation of what will really happen and there is nobody that can know for sure until it starts happening.
Keep in mind that Panama is surrounded and many times flooded with water anytime of the year. We are concscious of the risk of dengue and despite tremendous efforts of some peopler we haven´t been able to control dengue and Aedes Aegiptus. Somehow this idea seems to be fairly useful so we pray to have success in it. I have to say there are still a lot of careless people that continue to litter and who doesn´t care of their properties to avoid mosquitoes to reproduce.
Don’t mess with nature. The way to combat the mosquito is through better Higene and careful disposal of garbage. Need to eliminate water containers. Good thing that it is the dry season in Panama, otherwise the numbers of cases would be even higher!
Messing with nature is not generally a good idea. Hope Oxitech have got good insurance in case they get sued for an act of biological warfare.